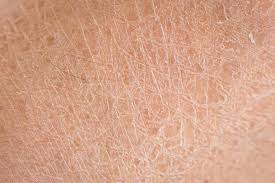
Pertinent Insights On The Relationship Between Footwear And Dry Skin
Unlike any other anatomical area, the feet are subject to much chafing, rubbing, and sweating, which can affect the skin surface. Wearing shoes without socks can dry out the feet depending on the material the shoes are made from. Due to body heat and moisture, there is almost always higher heat and humidity inside your shoes. If the shoe material is breathable as in leather, the heat and humidity being pushed out can escape from the shoes and such ventilation keeps your foot dry and comfortable. Non-breathable material such as vinyl blocks the heat and humidity inside. Some people have sweatier feet than others and this will interact with the shoe as well. Sweaty feet dry out the skin quicker. Shoes that do not fit as well (flip flops, open-backed sandals) produce more friction to dry out the skin. However, when dry skin occurs on the feet, the symptoms of discomfort are magnified due to shoe wear, the stretching of the skin on the feet each time we step down, and certain synthetic materials in the socks and shoes that dry the skin out even more. Due to the confining nature of the shoes we wear and the lack of fresh air that hits the skin of the feet due to our socks and shoes, dry feet need specialized care in order to prevent pain. In many cases, the shoes we wear can also protect our feet from dry skin and fissures if they fit properly and are made of breathable material.
Abnormal foot mechanics and deformities cause abnormalities in the way we walk. This subsequently causes certain areas of the feet to bear abnormal amounts of weight, which may result in dry patches, calluses, corns, and fissures. Orthotics and wearing the correct shoes help to spread out the abnormal weight and reduce friction. One must remember that the skin of the feet has no oil glands and must rely on the sweat glands to moisturize the skin. Sweat glands operate by secreting a substance comprised mostly of water, sodium chloride, and electrolytes. Accordingly, sweat is more “drying” than moisturizing. Each of our feet is densely covered with approximately 250,000 eccrine sweat glands, making feet one of the sweatiest places on the body.
The lack of oil glands makes preventing dry skin difficult but if we had oil glands on our feet, we would slip and slide with each step we took. Socks absorb sweat and are supposed to prevent blisters. It is known that certain synthetic socks can decrease the temperature of the foot as much as 3º and that is enough to prevent blister formation by limiting sweating. Dry feet are not the same as dry skin of the feet.  One study looked at fabric softeners and surmised that fabric softeners provide benefits to individuals with dry skin because of the decreased friction of the garments against the skin.12 Since friction results in heat, the heat will dry your feet out faster. It is known that nylon and rayon socks cause dryness to the skin. In one study, the prevalence of Type IV hypersensitivity to rubber allergens was evident in patients with stasis eczema and/or venous leg ulcers over an 18-month period.13 Accordingly, vascular hose in some patients may result in a dry, itchy skin response and when not wearing the hose at night, patients must moisturize.
One study looked at fabric softeners and surmised that fabric softeners provide benefits to individuals with dry skin because of the decreased friction of the garments against the skin.12 Since friction results in heat, the heat will dry your feet out faster. It is known that nylon and rayon socks cause dryness to the skin. In one study, the prevalence of Type IV hypersensitivity to rubber allergens was evident in patients with stasis eczema and/or venous leg ulcers over an 18-month period.13 Accordingly, vascular hose in some patients may result in a dry, itchy skin response and when not wearing the hose at night, patients must moisturize.
Treatment Tips For Specific Skin Conditions
Stucco keratosis. This is a keratotic papule that is usually present on the distal lower acral extremities of males. Stucco keratosis seems to appear with a higher frequency in males but it is not genetic. The lesion is asymptomatic and patients usually do not complain of having the lesions. The name stucco keratosis derives from the “stuck on” appearance of the lesions. The lesions are usually found in elderly patients. The differential diagnoses are seborrheic keratosis and melanoma.
Xerosis. Xerosis results in generalized or localized pruritus and dry, itchy skin. Rubbing and scratching causes increased irritation, leading to more pruritus and inflammation.14
Lichen simplex chronicus (neurodermatitis). Prolonged itching and scratching can lead to skin that is thick, scaly, and leathery. The patches can be raw, red, or darker than the rest of your skin.
Stasis dermatitis (venous eczema, varicose eczema). Stasis dermatitis is not “true” dermatitis but instead is due to venous hypertension in the lower leg. This is caused by insufficiency of the superficial veins as well as the long saphenous vein. One study found that when patients received classical flush ligation and a saphenectomy, lower leg dermatitis healed in all 10 patients within eight to 12 weeks, and there was no recurrence.15
Eczema craquelé (winter itch, asteatotic eczema, xerotic eczema, desiccation dermatitis). Eczema craquelé occurs mostly in elderly individuals with tight, red, dry skin that progresses to superficially fissured dermatitis. The irregular network of fissures resembles a dry riverbed and is visible on the shins of the legs. This is essentially “advanced” xerosis.9
Atopic dermatitis. In this form of eczema, one has more sensitive and drier skin due to an autoimmune condition.
Psoriasis. Psoriasis involves the rapid buildup of rough, dry, dead skin cells that form thick scales. These scales bleed when patients pick them off. It occurs mostly on the plantar skin of the foot.
Which Came First, Dry Skin Or The Itch?
Dry, “flaky” skin is an irritant and causes cutaneous inflammation, which in turn results in pruritus. Once the itch-scratch-itch cycle starts, the skin gets worse. The inflammation irritates the nerve endings.16 Treatment of dry skin is one of the most important measures against pruritus.17 Prevention is very important.
Researchers have rarely found correlations between itch and objective measures of barrier function and skin dryness such as skin hydration and transepidermal water loss. Recent experimental evidence indicates that damage to the stratum corneum with acetone/ether and water elicits a scratching response in mice and rats.18
Skin diseases associated with itch include eczema, atopic dermatitis, dry skin, contact dermatitis, psoriasis, lichen planus and bacterial infection.
Topical treatments for itch/dry skin: Polidocanol (Asclera, Merz Aesthetics) Capsaicin Menthol Phototherapy Lotions and creams Steroids Systemic treatments for itch/dry skin: Antihistamines Antidepressants Gabapentin (Neurontin, Pfizer) Opiate antagonist
Keys To Diagnosing Dry Skin
For the most part, dry skin is a purely clinical diagnosis. A thorough history, review of systems, and physical examination are critical to determining its cause. Examination of the skin may be misleading. There are frequently only secondary lesions, eczematous changes, lichenification, and excoriation, and the inciting cause may not be present. If primary lesions are present, a skin biopsy can lead to a diagnosis. One must consider systemic causes of dry skin/itch, such as cholestasis, uremia, hyperthyroidism, medications, or lymphoma.
However, if the normal topical treatments are not resolving the condition, have the lab test for thyroid function, renal function, liver function, HIV, zinc level, cancer, or Sjögren’s syndrome.
Treating Dry Skin
The first step to treat dry skin is to add water to the skin and apply a hydrophobic substance to keep it there. The substances include water-in-oil creams and lotions or 100% oil ointments to lock in the water. No matter what the cause of dry skin is, occlusive moisturizers, humectant emollients, and keratolytics are three commonly used topical treatments. For the most part, emollients work by retaining water in the skin where it is needed and enabling the repair of damaged cells on the skin’s surface. Emollients also act as a barrier to the environment, preventing irritants from penetrating the outer layer of the skin (epidermis) by creating a protective lipid film.3
Occlusive emollients such as petrolatum prevent water loss only by acting as a layer of oil on the surface of the skin to trap water and prevent evaporation. Humectant emollients such as Eucerin (Beiersdorf) penetrate the stratum corneum and draw water from the dermis to retain it in the epidermis.
Keratolytics such as lactic acid, salicylic acid, and glycolic acid help remove scales. Many times all three are together in one product.
While topical steroids do not treat dry skin, they do decrease the inflammation of the skin and the “itch” factor. Maximum hydration can occur with 60% propylene glycol in water applied under occlusion.19 When using topical steroids, choose a mid-potency steroid for dorsal skin and a high-potency to super-high-potency steroid on plantar skin. All topical steroids have increased absorption through the incomplete skin barrier.
When discussing the choice of emollients, a continuum exists between oily ointments and water-based creams and lotions. Ointments are best for the driest of skin conditions and are used at home when patients are not wearing tight clothes or working with others. The application of ointments can cause folliculitis in hairy areas, an unusual issue in the foot and ankle. The frequent use of emollients reduces the need for steroids.20
To avoid or treat xerosis, patients should moisturize their feet right after a bath or shower. They should avoid soaking their feet in hot water for long periods, using drying soaps on the feet, or scrubbing their feet dry. Anecdotal and limited data suggest that gabapentin, cutaneous field stimulation, serotonin antagonists, and ultraviolet B phototherapy may reduce itch in some of these patients.
Combating The Effects Of The Sun
The sun adds to the drying out of the skin just like the cool air of winter and the dry air of a desert. If the skin barrier is compromised, patients can have increased dryness. Snow, sand, and water increase the need for sunscreens because they reflect the sun’s rays. A sunscreen product acts like a very thin bulletproof vest, stopping the ultraviolet photons before they can reach the skin and inflict damage. It contains organic sunscreen molecules that absorb UV and inorganic pigments that absorb, scatter, and reflect UV. There are sunscreens now that help prevent dry skin by moisturizing as well.21
The sun’s heat dries out areas of unprotected skin and depletes the skin’s supply of natural lubricating oils. In addition, the sun’s UV radiation can cause burning and long-term changes in the skin’s structure. The sun causes dry skin, sunburn, and actinic keratosis. Skin appears dry, flaky, and wrinkled.
Other Considerations With Dry Skin Conditions
Cracked heels. Podiatrists frequently see cracked heels due to poor foot mechanics, prolonged standing, being overweight, poor shoe gear, open-backed shoes, and the usual cause, flip flops. It is the perfect storm of friction, lack of oil-producing glands and inadequate shoegear. In these conditions, the fat pad under the heel expands sideways and increases the pressure on the side. If there is any lack of moisture in the heel, it will crack. Accordingly, if you have an overweight patient with diabetes who wears house slippers much of the day, expect her to have cracks in the heels. Cracked heels are more common during the winter months although if you spend extended amounts of time in the sun, it can also dry out your skin to cause cracked heels. Medical conditions including diabetes, obesity, thyroid disease, and psoriasis may also cause dry, cracked heels.
Aged/elderly skin. In elderly skin, the epidermis thins, and the corneocytes do not adhere to each other as well and the skin loses its water-binding capacity. Dry skin is itchy skin and itchy skin is dry skin. It is difficult to split them up. Itching in the elderly presents a diagnostic and therapeutic challenge. A thorough history, review of systems, and physical examination are critical to determining the cause of itching. Examination of the skin may be misleading.
There are frequently only secondary lesions, eczematous changes, lichenification, and excoriation, which we may misdiagnose as primary dermatitis. Xerosis may be the cause but it is sometimes merely coincidental. If primary lesions are present, a skin biopsy can aid in diagnosis. Consider systemic causes of itching such as cholestasis, uremia, hyperthyroidism, medications, or lymphoma.
If the cause remains elusive, consider idiopathic itching of the elderly or so-called “senile pruritus.” The pathophysiology of this form of pruritus is poorly understood but, likely, age-related changes of the skin, cutaneous nerves, and other parts of the nervous system play a role.22
Treating Dry Skin That Is Concomitant With Common Diseases
Certain disease states can cause xerosis so in the workup, one should make note of congenital and acquired ichthyoses, atopic dermatitis, hypothyroidism, Down syndrome, renal failure, malnutrition, malabsorption, HIV, lymphoma, liver disease, Sjögren’s syndrome and certain drugs.
End-stage renal disease (ESRD). This is a progressive and irreversible kidney dysfunction lasting three months or more. Nearly all patients with ESRD have at least one dermatological disorder, and these skin and nail changes can occur before or even after initiation of dialysis or transplantation.23 Some authors have suggested that ESRD–associated xerosis may be a result of decreased water content in the epidermis. Clinical and histologic evaluations have shown an overall decrease in sweat volume in patients with uremia as well as the atrophy of sebaceous glands.24
Xerosis occurs in 50 to 75 percent of dialysis patients.24 It manifests as poor skin turgor with scaling, dryness and fissuring of the skin, particularly affecting extensor surfaces of extremities.
Of those with chronic renal failure, 15 to 49 percent experience pruritus. Of those patients undergoing dialysis, the prevalence is up to 50 to 90 percent but as dialysis has improved, it has become less common.24 In acute renal failure, pruritus is very uncommon. The pruritus of ESRD is most often generalized and light but can be severe and unremitting.23
Hypothyroidism. Hypothyroidism is more common in women and people over the age of 50. The thyroid produces too few thyroid hormones and this reduces the activity of the sweat and oil glands, leading to rough, dry skin. Symptoms with a high specificity for hypothyroidism include constipation, cold intolerance, proximal muscle weakness, hair thinning, and dry skin. Dry skin can be a symptom of hypothyroidism in infants and children as well. A blood test can determine the level of thyroid stimulating hormone as well as thyroid hormones T3 and T4.
Diabetes. People with diabetes have a high incidence of xerosis of the feet, especially on the heels. While assessing for predictors of foot lesions in patients with diabetes, one study found that 82.1 percent of their patients had skin with dryness, cracks, or fissures.25 An unpublished survey of 105 consecutive patients with diabetes conducted by one of the authors revealed that 75 percent had clinical manifestations of dry skin. Dry skin often leads to cracks and fissures, which can serve as a portal of entry for bacteria.
Despite possible “dry skin” definition discrepancies across the studies, it is clear that skin dryness is one of the earliest and most common manifestations of type 1 diabetes. The clinical observations are supported by objective findings of a reduced hydration state of the stratum corneum and decreased sebaceous gland activity in patients with diabetes without any impairment of the stratum corneum barrier function.26
Liver disease. The liver neutralizes toxins and filters bile salts. If the liver’s function is impaired, these materials can accumulate in the body, and deposition in the skin causes irritation and itching. In cholestatic liver disorders such as primary sclerosing cholangitis and obstructive gallstone disease, pruritus tends to be generalized but is worse on the feet and hands.27
In Conclusion
Dry skin can be persistent and recurring due to the long list of possible causes. Clinicians often treat dry skin with hydrophilic and/or lipophilic moisturizers.
Hydrophilic moisturizers must penetrate the stratum corneum deeply to function properly whereas lipophilic moisturizers should remain in the upper stratum corneum layers.28
Traditionally, clinicians used humectant and occlusive technologies to treat dry skin. Originally, non-lamellar forming ingredients such as petrolatum were in use but recent research has shown an advantage of using lamellar-forming factors such as ceramides, pseudoceramides, and phospholipids.29
As with all topical treatments, adherence is the great challenge one faces in the management of skin diseases. Strong odor from ingredients and greasy compositions may be disagreeable to the patients. Furthermore, low pH and sensory reactions, from lactic acid and urea, for example, may reduce patient acceptance.30
The number of studies on skin barrier function and hydration is endless. There is a long list of products available and some may work better depending on certain skin characteristics of the person. Many podiatry-friendly companies have products that include CeraVe (Valeant), Eucerin, AmLactin (Upsher Smith), Cetaphil (Galderma), Borage Therapy (ShiKai), Uramaxin (Medimetriks), Carmol 40, Lubriderm (Johnson and Johnson), and Aveeno (Johnson and Johnson).
Dr. Morse is the President of the American Society of Podiatric Dermatology. He is a Fellow of the American College of Foot and Ankle Surgeons, and the American College of Foot and Ankle Orthopedics and Medicine. Dr. Morse is board-certified in foot surgery. He is on the Podiatric Residency Educational Committee at the MedStar Washington Hospital Center in Washington, D.C.
References
1. Nolan K, Marmur E. Moisturizers: Reality and the skin benefits. Dermatologic Therapy. 2012; 25(3):229-233.
2. Coderch L, López O, de la Maza A, Parra JL. Ceramides and skin function. Am J Clin Dermatol. 2003; 4(2):107-29.
3. Watkins P. Using emollients to restore and maintain skin integrity. Nursing Standard. 2008; 22(41):51-57.
4. Pons-Guiraud A. Dry skin in dermatology: a complex physiopathology. J Eur Acad Dermatol Venereol. 2007; 21(Supp 2):1-4.
5. Vorgeli D. The vital role of emollients in the treatment of eczema. Br J Nursing. 2011; 20(2):74-80.
6. Choi MJ, Maibach HI. Role of ceramides in barrier function of healthy and diseased skin. Am J Clin Dermatol. 2005; 6(4):215-23.
7. Johnsen G, Haugsnes A. A new approach for an estimation of the equilibrium stratum corneum water content. Skin Research Technology. 2010; 16(2):142-145.
8. McKinley-Grant L. VisualDx: Essential Dermatology in Pigmented Skin. Lippincott, Philadelphia, 2011, pp. 322.
9. Aziz N. Xerosis and eczema craquele. In McKinley-Grant L (ed): VisualDx: Essential Dermatology in Pigmented Skin. Lippincott, Philadelphia, 2011, pp. 316.
10. Johnson B, Moy R, White G. Ethnic Skin – Medical and Surgical. Mosby, St Louis, 1998, p. 4.
11. Jiang ZX. DeLaCruz J. Appearance benefits of skin moisturization. Skin Research & Technology. 2011; 17(1):51-5.
12. Fujimura T, Takagi Y. Real-life use of underwear treated with fabric softeners improves skin dryness by decreasing the friction of fabrics against the skin. Int J Cosmet Sci. 2011;33(6):566-571.
13. Gooptu C, Powell SM. The problems of rubber hypersensitivity (Types I and IV) in chronic leg ulcer and stasis eczema patients. Contact Dermatitis. 1999; 41(2):89-93.
14. Arndt K, Hsu J. Manual of Dermatologic Therapeutics, seventh edition. Wolters Kluwer, Philadelphia, pp. 72-74.
15. Sippel K, Mayer D, Ballmer B, Dragieva G, Lauchli S, French LE, Hafner J. Evidence that venous hypertension causes stasis dermatitis. Phlebology. 2011; 26(8):361-5.
16. Hazin R. Recognizing and treating cutaneous signs of liver disease. Cleve Clin J Med. 2009; 76(10):599-606.
17. Bigliardi PL. Pruritus–causes, diagnostics and treatment. Revue Medicale Suisse. 2006; 2(63):1115-8.
18. Yosipovitch, G. Dry skin and impairment of barrier function associated with itch – new insights. J Cosmet Sci. 2004; 26(1):1-7.
19. Scott S. Atopic dermatitis and dry skin. In: Krinsky D. Berardi R, Ferreri S, et al (eds). Handbook of Nonprescription Drugs, 17th edition. American Pharmacists Association, Washington, DC, 2012, pp. 615-630.
20. Domino F. 5-Minute Clinical Consult 2014, 22nd edition. Lippincott Williams and Wilkins, Philadelphia, 2013.
21. Available at http://www.scientificamerican.com/article.cfm?id=how-does-sunscreen-protec . Published May 7, 2007. Accessed Dec. 6, 2013.
22. Ward JR, Bernhard JD. Willan’s itch and other causes of pruritus in the elderly. Int J Dermatol. 2005; 44(4):267-273.
23. Lynde C, Kraft J. Skin manifestations of kidney disease: Conditions range from benign to life-threatening. Parkhurst Exchange. 2007; Vol.15, No.02
24. Nunley JR, Elston DM. Dermatologic manifestations of renal disease. Available at http://emedicine.medscape.com/article/1094846-overview . Published April 11, 2012. Accessed Dec. 9, 2013.
25. Litzelman DK, Marriott DJ, Vinicor F. Independent physiological predictors of foot lesions in subjects with NIDDM. Diabetes Care. 1997; 20(8):1273-1278.
26. Pavlović MD. The prevalence of cutaneous manifestations in young patients with type 1 diabetes. Diabetes Care. 2007; 30(8):1964-1967.
27. Ballmer-Weber BK, Dummer R. Pruritus in frequent skin diseases and therapeutic options. Praxis. 2007; 96(4):107-11.
28. Caussin J, Rozema E, Gooris GS, Wiechers JW, Pavel S, Bouwstra JA. Hydrophilic and lipophilic moisturizers have similar penetration profiles but different effects on SC water distribution in vivo. Experimental Dermatol. 2009; 18(11):954-61.
29. Pennick G, Chavan B, Summers B, Rawlings AV. The effect of an amphiphilic self-assembled lipid lamellar phase on the relief of dry skin. Int J Cosmet Sci. 2012; 34(6):567-74.
30. Loden M. Role of topical emollients and moisturizers in the treatment of dry skin barrier disorders. Am J Clin Dermatol. 2003; 4(11):771-88.
Editor’s note: For related articles see “Keys To Differentiating Eczematous Eruptions In The Pedal Skin” in the April 2009 issue of Podiatry Today, “What You Should Know About Atopic Dermatitis” in the September 2005 issue, “A Guide To Skin Conditions Of The Diabetic Foot” in the September 2004 issue or “Treating Psoriasis In The Lower Extremity” in the February 2011 issue.
To access the archives, visit www.podiatrytoday.com. For an enhanced online experience, check out Podiatry Today on your iPad or Android tablet.